 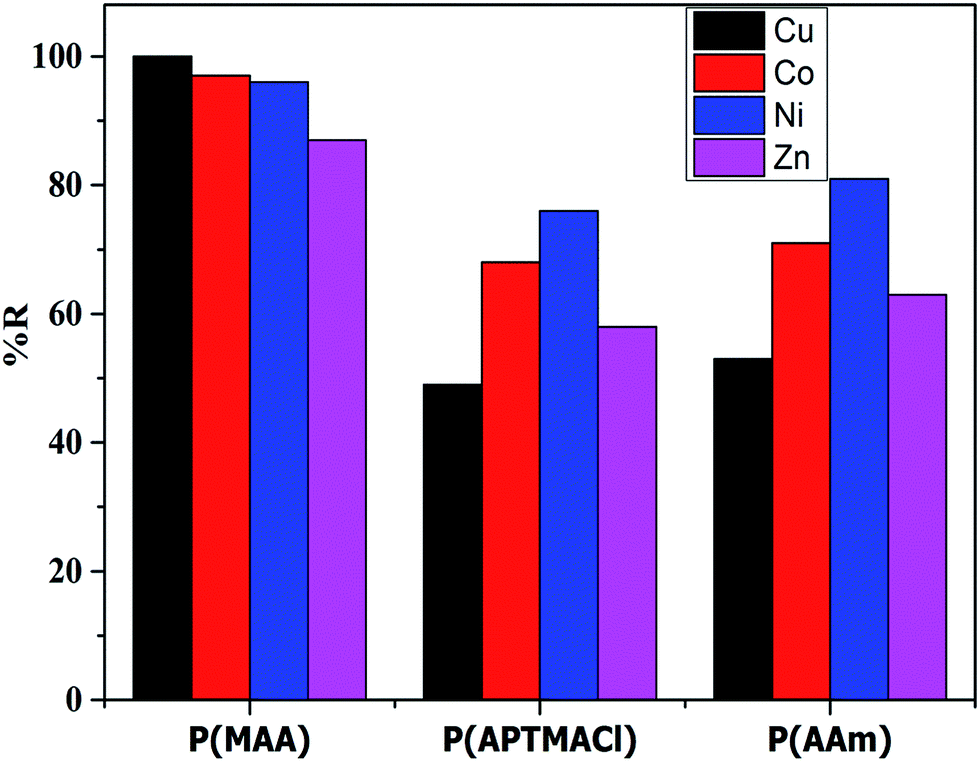
Other strategies, such as those based on core–shells, concentration gradients, and single-crystal structures, are only incidentally mentioned and considered undeveloped concepts. Most reviews have mainly focused on the failure of layered oxides caused by electrochemical cycling and storage along with the two conventional methods of coating and atomic doping. Many researchers have published excellent reviews detailing the research progress on understanding critical challenges that such cathode materials and corresponding strategies face to fulfill the goal of high energy density. Even so, next-generation batteries with nickel-rich layered oxide cathodes and graphite–silicon anodes are still expected to achieve cell-level specific energy densities approaching 350 Wh kg −1 by 2025 or earlier. In particular, nickel-based cathodes ( \(\geqslant\)80% nickel content) with high electrode densities that exceed ≈ 3.3 g cm −3 usually undergo structural collapse, exhibiting severe capacity fading as the gravimetric energy density increases. However, although the high proportion of nickel in the family of nickel-based layered oxide materials can allow for increased gravimetric energy, the low reversible capacity of layered oxides with high nickel contents hinders their further application in commercial batteries. Nickel-based oxides LiNi 1– x– 圜o xAl yO 2 (NCA) and LiNi 1– x– 圜o xMn yO 2 (NCM) are obtained by introducing cobalt and manganese (aluminum) into LiNiO 2 to improve its structural and thermal stability. Delithiated LiNiO 2 easily releases oxygen even under normal operating conditions and undergoes exothermic reactions with organic electrolytes. Nevertheless, high-quality LiNiO 2 samples are very difficult to synthesize and prone to chemical and structural instability, particularly at high cutoff voltages. LiNiO 2, which has the same layered structure as LiCoO 2, was first considered as an alternative cathode material given the high cost and scarce resources of elemental cobalt. Lithium cobalt oxides continue to play a dominant role among many positive materials. Since then, after being introduced in the first commercial lithium-ion battery (LIB, energy density: ~ 150 Wh kg −1) by Sony, lithium cobalt oxides have been utilized in batteries as cathode materials. In 1981, the Goodenough group first discovered that lithium cobalt oxides (LiCoO 2) had high conductivity and maintained their structural stability throughout ≈ 0.5 Li + (de)intercalation per formula unit. Future development should focus on facile production with strong control of the particle size and distribution, structural defects, and impurities to fully reap the benefits of single-crystal materials. Industrial techniques and facilities are also reviewed in combination with our group’s experiences in single-crystal research. The synthesis science behind single-crystal materials and comparative studies between single-crystal and polycrystal materials are discussed in detail. This review highlights origins, recent developments, challenges, and opportunities for single-crystal layered oxide cathodes. A comprehensive understanding of the growing body of research related to single-crystal materials is imperative to improve the performance of cathodes in LIBs. Single-crystal materials, which exhibit robust mechanical strength and a high surface area, have great potential to address the challenges that hinder their polycrystal counterparts. These phenomena accelerate cell degradation. 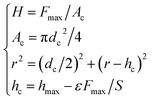
However, polycrystal particles tend to suffer from pulverization and severe side reactions along grain boundaries during cycling. Traditional nickel-based oxide particles, usually called polycrystal materials, are composed of microsized primary particles. Nickel-rich layered oxides have largely replaced LiCoO 2 in commercial batteries because of their low cost, high energy density, and good reliability. Lithium-ion batteries (LIBs) represent the most promising choice for meeting the ever-growing demand of society for various electric applications, such as electric transportation, portable electronics, and grid storage. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
